
A simple blood test that will be trialled in Ballarat could spare cancer patients unnecessary chemotherapy and its often-debilitating side effects.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
Ballarat Regional Integrated Cancer Centre is one of 40 hospitals in Australia and New Zealand taking part in a trial to investigate if a simple blood test can determine whether or not patients need chemotherapy after cancer surgery.
Many cancer patients receive chemotherapy after surgery as a precaution because there is no reliable way to determine which patients will suffer a recurrence of their cancer after surgery.
It is hoped the blood test, developed in Melbourne at the Walter and Eliza Hall Institute of Medical Research, will prevent patients at low risk of a second bout of cancer from receiving unnecessary chemotherapy.
It’s a very interesting trial and it’s great to be able to give this opportunity to patients in Ballarat.
- BRICC oncologist Dr Geoffrey Chong
Results from the blood test could also help doctors scale the dose of chemotherapy for individual patients based on their risk of the cancer returning.
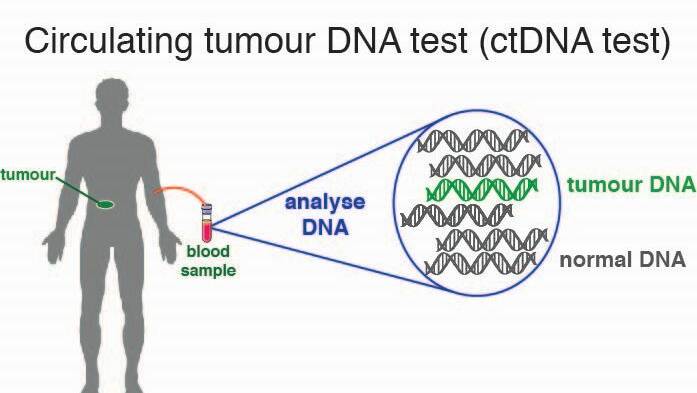
WEHI clinician-researcher Associate Professor Sumi Ananda said the blood test was looking for fragments of circulating tumour DNA in a patient’s blood after the tumour had been surgically removed.
Trials began in bowel cancer patients in 2015 and results have shown it can determine whether patients can be divided in to high risk and low risk groups. The trial was extended to women with ovarian cancer in 2017 and also shows promise in pancreatic cancer.
“Eighty per cent of bowel cancer patients have tumours that appear to be limited to the bowel, but we don’t known in which patients it will and won’t recur,” Assoc Prof Ananda said.
“Examination is done by pathology of the tumour when it is removed at the time of surgery, specifically looking whether it is high risk … but some high risk patients won’t have a recurrence because it has been cured by surgery, and low risk patients may suffer from recurrence so many patients are treated with six months of chemotherapy even though they may not need it, and others that will potentially benefit from treatment won’t get chemo because they appear to be too low risk.
The blood test can detect minute amounts of the tumor DNA and the current trials will investigate how or whether the blood test can improve cancer patient management.
Assoc Prof Ananda said results so far had shown the risk of recurrence was above 80 per cent for those with detectable circulating tumour DNA and just 8 per cent for those without.
The blood test trial, developed in collaboration between WEHI and the Johns Hopkins Kimmel Cancer Centre in the US, will determine whether the amount of tumour DNA in a patient’s blood indicates the risk of relapse.
The US arm of the trial is looking to see whether the blood test could be used for population screening to detect early stage bowel cancer in the population, similar to the current National Bowel Cancer Screening Program.
Hopefully we will identify a group of patients who will not require chemo at all, in which case they wouldn’t be exposed to any side effects.
- BRICC oncologist Dr Geoffrey Chong
BRICC is taking part in stage two trials of the blood test for people with newly diagnosed bowel cancer with lymph gland involvement.
“It’s a very interesting trial and it’s great to be able to give this opportunity to patients in Ballarat,” said BRICC oncologist Dr Geoffrey Chong.
BRICC was initiatied as a trial site this week and will enroll eligible patients as they come through.
“We know a proportion of patients have microscopic secondary tumours that have already spread beyond the surgical field that can’t be detected on scans or current blood tests.”
Eligible patients will have a blood test four or five weeks after their surgery, then randomised in to treatment groups.
Dr Chong said he saw about 20 patients a year for chemotherapy following bowel cancer surgery at BHS.
Dr Chong said the blood test could potentially see treatments tailored depending on risk.
“If it is positive those patients could get increased, escalated type of chemotherapy with an extra drug and if their tumour DNA is negative they they could get de-escalated or shorter chemotherapy or potentially no chemo at all,” he said.
The goal is to spare patients from undergoing chemotherapy if they do not need it.
People who are receiving chemotherapy may experience a range of side-effects including pain, fatigue, nausea and other digestive issues, bleeding problems and an increased susceptibility to infection. Long term side-effects of chemotherapy may include heart, lung, nerve and memory problems, and fertility issues.
“Hopefully we will identify a group of patients who will not require chemo at all, in which case they wouldn’t be exposed to any side effects,” Dr Chong said.
RELATED STORY: Tackling Ballarat’s bowel cancer test rate
The study is hoping to recruit 2000 patients across the 40 sites making it one of the largest trials in Australia and internationally to investigate a predictive blood test to guide cancer treatment. The trials are expected to run until 2021 for bowel cancer and 2019 for ovarian cancer.
Ballarat resident Jessica finished six months of chemotherapy in July after being diagnosed with bowel cancer and undergoing surgery late last year.
“I was quite fortunate with my chemo treatment as … I didn’t lose my hair, was able to continue to work part time, and caring for my family and doing daily life,” she said.
“Chemotherapy was my only option and with my knowledge of what chemo was I was petrified and would have been so excited to have thought a simple blood test could tell whether I needed to have it,” she said.
“Because you have already had thousands of blood tests, having a blood sample taken is no biggie … and to not have to go through chemo would be quite exciting.”
Have you signed up to The Courier's daily newsletter and breaking news emails? You can register below and make sure you are up to date with everything that's happening in Ballarat.

